Published On Feb 3, 2023
The first in a four-part series on intermolecular (or interparticle) forces.
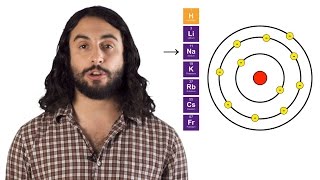
Polarizability is the tendency of an electron cloud in an atom or molecule to be distorted. This is explained with the examples of an atom's and molecule's electron cloud.
#chemistry
#polarizability
#Polarisability
Other videos in the lecture series
Dispersion Forces - • Dispersion Forces - London Forces - v...
Dipole-Dipole - • Dipole - Dipole Intermolecular Forces...
Hydrogen Bonding - • Hydrogen Bonding Intermolecular Force...
Dipole-Dipole and Hydrogen Bonding (Full Lecture) - • Dipole - Dipole and Hydrogen Bonding ...
Ionic Bonding - • Ionic Bonding Explained